|
I would encourage you to explore further. looking for relationships in a meaningful way. Bohrs model suggests that each atom has a set of unchangeable energy levels, and electrons in the electron cloud of that atom must be in one of those energy levels. It was already known that when a charged particle (such as an electron) moves in a curved path, it gives off some form of light and loses energy in doing so. 
Unfortunately, there was a serious flaw in the planetary model. They also help us explain and predict the behavior of atoms. 9.4: The Bohr Model - Atoms with Orbits is shared under a CK-12 license and was authored, remixed, and/or curated by Marisa Alviar-Agnew & Henry Agnew. 3: Niels Bohr with Albert Einstein at Paul Ehrenfests home in Leiden (December 1925). Models help us visualize atomic structure. As a theory, it can be derived as a first-order approximation of the hydrogen atom using the broader and much more accurate quantum mechanics and thus may be considered to be an obsolete scientific theory. Atoms are way too small to see with the naked eye (and even most microscopes). The Bohr model is a relatively primitive model of the hydrogen atom, compared to the valence shell model. We can, however, say that the more electrons there are in an atom, the greater the variety of photon energies there will be so this may be a link worth exploringĪs I say, you are asking a great question. The Bohr model Google Classroom Learn how Bohr models are used to represent atoms. as it switches from one energy level to another, then it emits / absorbs photons. This is because the electron can exist in many energy levels. Hydrogen, for example, although the simplest atom, has a whole range of photon energies that it emits. The value of the energies of photons does depend on the available energy levels in the atom. Now, the energy of the photon emitted from any element does not depend on the number of electrons in the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called electrons, circulate at some distance. Every circular orbit will have a certain amount of fixed energy and these circular orbits were termed orbital shells. The orbits are termed as stationary orbit. The Bohr model is a relatively primitive model of the hydrogen atom, compared to the valence shell model. 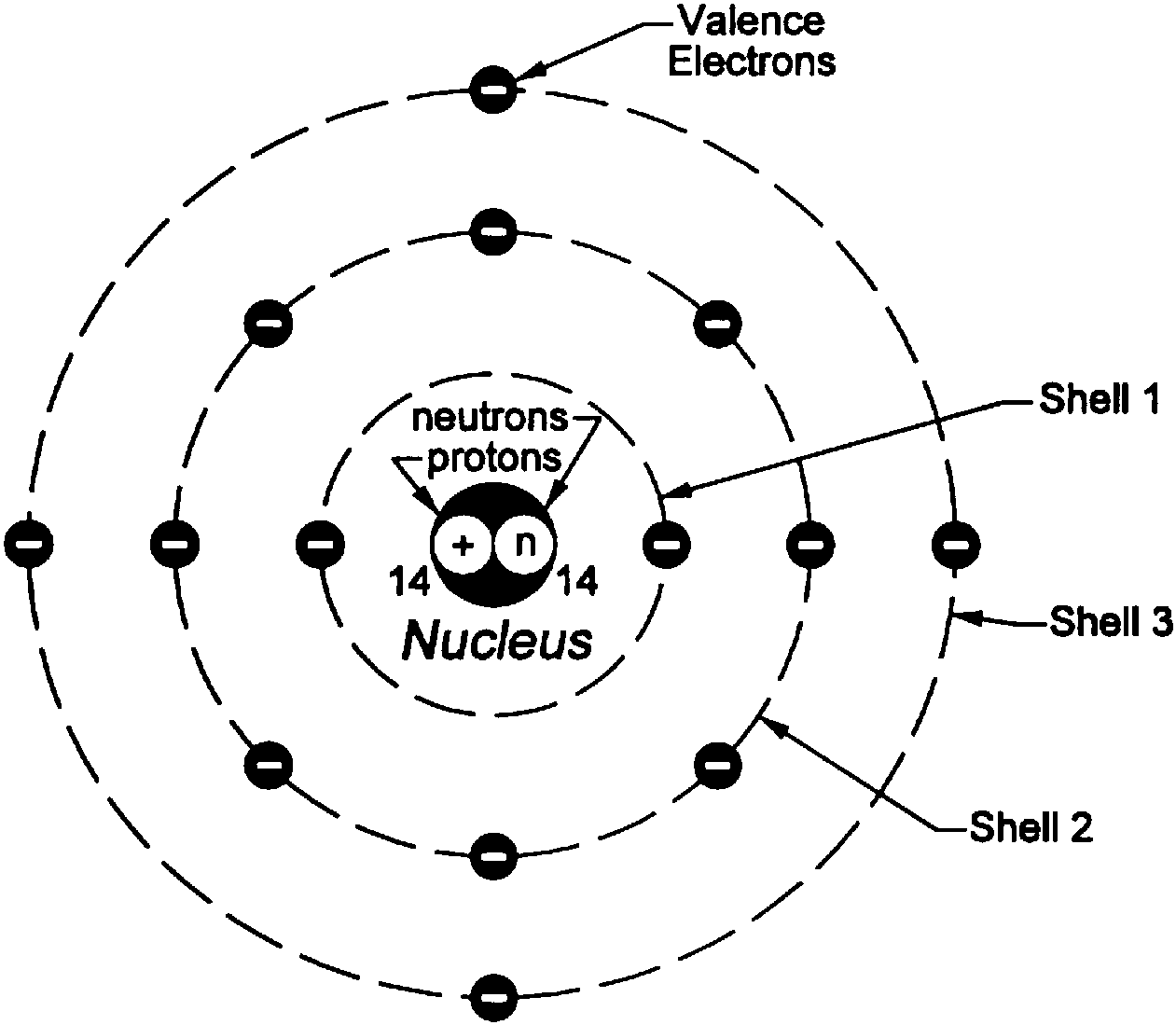
The electronic configuration of Oxygen is He 2s 2 2p 4. The information that we can derive from the above-mentioned Oxygen box is as follows: The atomic number of Oxygen is 8. OK: I would say that the periodic table tells us about the number of protons in an element and, therefore the number of electrons too. Rutherford model, description of the structure of atoms proposed (1911) by the New Zealand-born physicist Ernest Rutherford. Electrons revolve around the nucleus in a fixed circular path termed orbits or shells or energy level. The oxygen atom belongs to the 16 th group of the periodic table. Here is my answer, but I would encourage you to explore this and similar questions further.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
